Evaluation of Shawn Mikula’s whole mouse brain entry
Dr. Shawn Mikula is a neuroscientist currently at the Max Planck Institute of Neurobiology in Martinsried, Germany. Dr. Mikula is leading their Whole Mouse Brain Volume Electron Microscopy Project with the incredibly ambitious goal to map the entire connectome of a mouse brain. Over the last five years he has been painstakingly researching modifications to the “standard EM” chemical fixation and staining protocol used for decades by electron microscopists. As is well known to all electron microscopists, the traditional protocol for preparing brain tissue for EM imaging only works for small pieces of tissue. The key problem has been that the chemicals used to preserve (and stain) the lipid membrane of cells, a mixture of osmium tetroxide with potassium ferrocyanide, is prone to precipitation and barrier formation within the tissue. This has limited high-quality ultrastructure preservation and staining to depths of just a few hundred microns thick.
Dr. Mikula has developed a protocol that overcomes this limitation. The trick appears to be to add a high concentration of formamide to the osmium tetroxide fixative. According to his recent paper an aqueous solution containing 40 mM OsO4, 35 mM K4Fe(CN)6, 100 mM Cacodylate buffer (pH 7.4) and 2.5 M formamide is sufficient to completely eliminate barrier formation allowing for uniform preservation and staining of an entire mouse brain. Dr. Mikula has recently published this protocol in the prestigious journal Nature Methods, a result significant enough to warrant him the cover image of the June 2015 issue.
Dr. Mikula has been an official competitor in our Brain Preservation Prize competition since 2012. In that time he has sent us many electron micrographs of his whole mouse brain tests demonstrating promising results. On three separate occasions in the past, Dr. Mikula has provided the BPF with a mouse brain for official evaluation, however on both of those occasions we found damage sufficient to reject the entry. The first brain we imaged showed large cortical cracks which were revealed when we x-ray imaged it. In the second brain, osmium penetration was not uniform leaving core regions like the thalamus insufficiently preserved. Dr. Mikula’s paper discusses the crack issue and its likely causes and solutions.
Large-scale evaluation of brain preservation by X-ray micro CT scan:
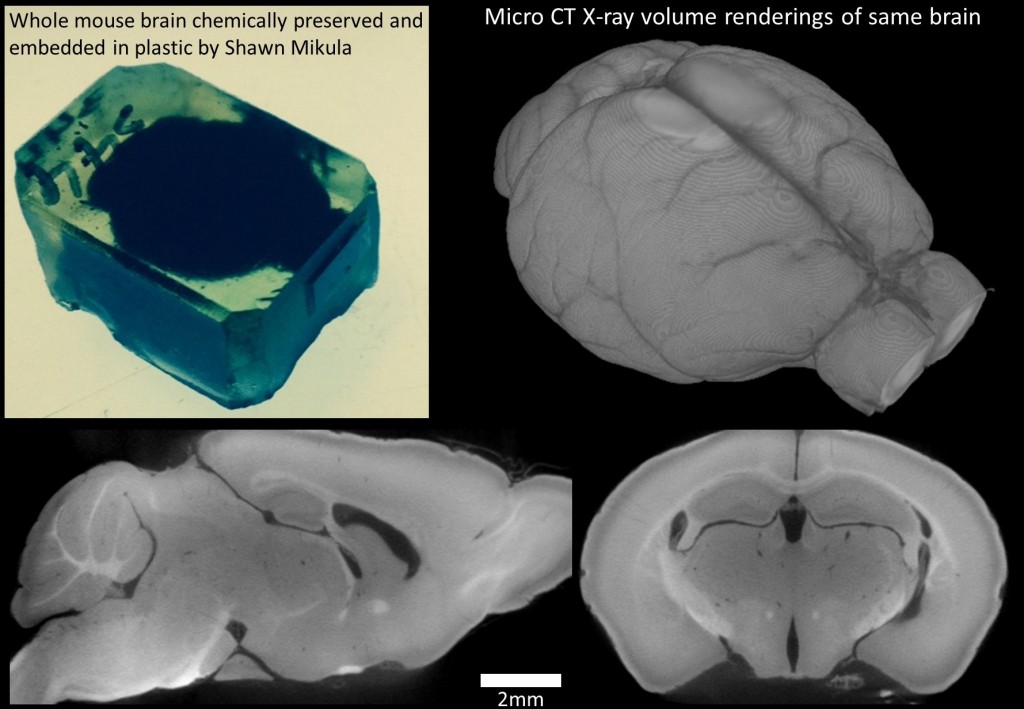
Below is a picture of the third, chemically preserved, plastic embedded whole mouse brain Dr. Mikula sent to us as an official entry in the small mammal phase of our Brain Preservation Prize competition. The chemical used to preserve the lipid membranes of cells (osmium tetroxide) darkens the tissue to deep black in color which is why the brain looks so dark in its plastic block. As a preliminary check of quality we performed an X-ray micro CT scan on the brain with a voxel resolution of 16 microns. The results of our X-ray volume imaging are shown below. Aside from minor dissection damage (which is to be expected when a brain is removed from the skull) the brain appears undamaged and to have relatively uniform staining at this scale of imaging:
Whole brain electron micrograph statistical survey for ultrastructure damage:
A crucial part of our evaluation process is a statistical survey looking for signs of ultrastructure damage. We use a precision diamond circular saw to chop 2mm thick coronal sections (slabs) off of the brain block. These are individually epoxied to metal SEM studs. We built a jig to hold a mounted slab in the chuck of an ultramicrotome so that its surface could be diamond polished. The section is then coated with a 100nm layer of gold (to prevent charging in the SEM) and mounted on the ultramicrotome again for final polishing with a very high quality diamond knife. Finally, 3nm of carbon is vacuum deposited on this polished surface (again to prevent charging). Here is an overview of the process:
This procedure allows us to acquire 2D electron micrographs of any region of the polished coronal section, and to relate these images back to the exact location in the original brain by correlating overview EM images with the full X-ray volume.Here is a subset of the 2D evaluation images we have collected so far on the Mikula entry. Please click on the small thumbnail images below to go to the full image (each full image is 4MB):